
Perspectives on sexual transmission of Ebola virus: complementarity between research and public health intervention
In the history of Filovirus outbreaks, sexual transmission was described in 1967 during the very first Marburg outbreak in Germany (1). And few years later, in 1990, a case of relapse following a lab exposure was reported in Novosibirsk, Russia (2). The analysis of the origin of the 23 Ebola outbreaks that occurred between 1976 and 2012 shows that for many events the exposure of the initial cases was not elucidated (Table 1). For two events (in italic in Table 1) in 1977 (Tandala, DRC) and 2011 (Luwero, Uganda) the index cases were respectively 14- and 12-year old girls for whom the mode of infection was never elucidated despite intensive investigations.
Table 1
| Year | Country | Species | Origin | Type |
|---|---|---|---|---|
| 1976 | DRC | Zaire | Antelope? | ? |
| 1976 | Sudan | Sudan | Bats? Or hunting party? | ? |
| 1977 | DRC | Zaire | Unknown | UNK |
| 1979 | Sudan | Sudan | Unknown | UNK |
| 1994 | Côte d’Ivoire | Tai Forest | Chimpanzee | Animal |
| 1994 | Gabon | Zaire | Chimpanzee, Gorilla | Animal |
| 1995 | DRC | Zaire | Chimpanzee | Animal |
| 1996 | Gabon | Zaire | Chimpanzee | Animal |
| 1996 | Gabon | Zaire | Chimpanzee | Animal |
| 2001 | Uganda | Sudan | Unknown | UNK |
| 2001 | Gabon | Zaire | Gorilla, Chimpanzee, Duiker | Animal |
| 2002 | Congo | Zaire | Gorilla, Chimpanzee, Monkey | Animal |
| 2003 | Congo | Zaire | Gorilla, Duiker | Animal |
| 2003 | Congo | Zaire | Monkey, Duiker | Animal |
| 2004 | Sudan | Zaire | Monkey | Animal |
| 2005 | Congo | Zaire | Wildlife (Monkey, Elephant) | Animal |
| 2007 | DRC | Zaire | Fruit Bats | ? |
| 2007 | Uganda | Bundibugyo | Bats? | ? |
| 2008 | DRC | Zaire | Unknown | UNK |
| 2011 | Uganda | Sudan | Unknown | UNK |
| 2012 | Uganda | Sudan | Unknown | UNK |
| 2012 | DRC | Bundibugyo | Unknown | UNK |
| 2012 | Uganda | Sudan | Unknown | UNK |
?, not fully elucidated.
Human-to-human transmission of Ebola virus (EBOV) happens primarily through direct contact with acutely sick patients, or with bodies of individuals who have died of Ebola virus disease (EVD). EBOV can be transmitted through broken skin or mucous membranes from the blood, body fluids, and secretions of the infected person (3,4) while aerosol transmission has not been documented (5). The first occurrence of possible Ebola acquired through sexual transmission was reported during November 2014 in Liberia. Several EVD flare-ups were subsequently confirmed to be due to exposure to survivors’ body fluids, and men semen testing programmes where set-up in the three most affected countries like the one reported from Liberia (6).
The EVD outbreak that started in West Africa in December 2013 has had dramatic consequences for the three most affected countries, Guinea, Liberia and Sierra Leone, with 28,616 confirmed, probable, and suspected cases of EVD, including 11,310 reported deaths (5). Although estimates of the exact number of people affected during the outbreak vary, it is probable that there are up to 10,000 EVD survivors among the three countries.
Persistence of Ebola virus in semen
Although sexual transmission was suspected before this outbreak, little was known about the persistence of EBOV in body fluids and in particular in semen. A literature review performed in May 2015 (7), identified only five original articles and one case report presenting results of any kind on persistence of EBOV in individuals who had survived acute EVD and recovered. Altogether, as of May 2015, persistence of the virus in seminal fluids had been documented in 11 patients.
In March 2015, a new flare of EVD due to exposure to infected survivor body fluids was reported in Liberia (8). By early 2015, several research groups initiated studies in Guinea, Liberia and Sierra Leone, to explore the duration of persistence of EBOV in body fluids and in particular in semen. The baseline data of the study in Sierra Leone was published in October 2015 (9) and showed that EBOV RNA identified by RT-PCR could be detected for as long as 9 months after onset of symptoms (Figure 1). Recent results (not yet published) indicate that EBOV RNA can persist for much longer in semen.
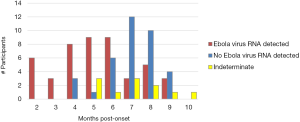
On the basis of preliminary results of persistence studies, WHO advised in June 2015 the three Member States most concerned by the epidemic to implement national semen testing programmes to be able to follow-up survivors, provide adequate counselling and care, and prevent further transmission through semen (10).
A national testing programme was set up in Liberia in this context. The article by Soka et al. (6) describes the implementation of this programme. With full respect for the principles of human rights, confidentiality and respect for participants, male survivors were enrolled in the programme on a voluntary basis and semen was collected until two consecutive specimen, collected at minimum 2 weeks apart, were found negative. Two negative consecutive semen sample, collected at minimum 1 week interval, were subsequently proposed by WHO as standard procedure, which was adopted by the three countries to consider a male survivor Ebola virus free (10).
The article published in Lancet Global Health (6) describes the preliminary results of the information collected from participants while enrolled in the programme, as well as behavioural outcomes.
The report focuses on 429 survivors. The longest interval between discharge from an Ebola treatment unit and a RT-PCR positive results was 565 days, and survivors older than 40 years were more likely to have at least one semen sample testing positive by RT-PCR than survivors younger than 40. In terms of public health, one of the most important results of this testing programme was that the survivors trusted the programme and would recommend participation to other survivors. This demonstrates that such national programmes are not only needed to prevent new flares of EVD and potentially new outbreaks, but also to address survivors’ concerns about their chances to transmit the virus and to fulfil their right to this information.
Few important points need to be highlighted: (I) the importance of the involvement of survivors’ associations, not only to engage participants in these national programmes, but also to ensure that communication around the results are translated and conveyed correctly; (II) counselling of the survivors according to their results: special counselling scripts were developed related to the meaning and consequences of a positive RT-PCR result, with common methods and messages across the three countries national programmes and related research studies. Increased correct condom use among survivors who received counselling demonstrate the importance of counselling; (III) the results are based on RT-PCR and therefore do not inform on the infectiousness of the virus.
This programme, together with those implemented in the other two countries, as well as the research projects conducted on the persistence of EBOV in body fluids, are crucial elements tackle EVD, not only because they provide essential information on the duration of virus persistence, but also because they have contributed to the decrease of stigma vis a vis survivors. Indeed, adequate counselling and information empowered survivors to discuss with their family, in some cases with their community, leading to reduced stigma.
Finally, these testing programmes and associated research studies have been instrumental in increasing our understanding of the virus, which will permit a quicker and more adapted response in the event of a new outbreak. In addition, this successful collaboration between outbreak response actors, national Sexual and Reproductive Health programmes, and Research and Development partners, has paved the way for similar synergies to address the Zika crisis and associated congenital brain abnormalities, including microcephaly, and Guillain-Barré syndrome (GBS).
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Journal of Public Health and Emergency. The article did not undergo external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jphe.2016.12.15). The authors are staff members of the World Health Organization. The authors alone are responsible for the views expressed in this article, which do not necessarily represent the decisions, policy or views of the World Health Organization.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Martini GA, Schmidt HA. Spermatogenic transmission of the "Marburg virus". (Causes of "Marburg simian disease"). Klin Wochenschr 1968;46:398-400. [Crossref] [PubMed]
- Nikiforov VV, Turovskiĭ IuI, Kalinin PP, et al. A case of a laboratory infection with Marburg fever. Zh Mikrobiol Epidemiol Immunobiol 1994;104-6. [PubMed]
- To KK, Chan JF, Tsang AK, et al. Ebola virus disease: a highly fatal infectious disease reemerging in West Africa. Microbes Infect 2015;17:84-97. [Crossref] [PubMed]
- Osterholm MT, Moore KA, Kelley NS, et al. Transmission of Ebola viruses: what we know and what we do not know. MBio 2015;6:e00137 [PubMed]
- Situation Report Ebola Virus Disease 10 June 2016, World Health Organization, Geneva, Switzerland. Available online: http://apps.who.int/iris/bitstream/10665/208883/1/ebolasitrep_10Jun2016_eng.pdf?ua=1
- Soka MJ, Choi MJ, Baller A, et al. Prevention of sexual transmission of Ebola in Liberia through a national semen testing and counselling programme for survivors: an analysis of Ebola virus RNA results and behavioural data. Lancet Glob Health 2016;4:e736-43. [Crossref] [PubMed]
- Thorson A, Formenty P, Lofthouse C, et al. Systematic review of the literature on viral persistence and sexual transmission from recovered Ebola survivors: evidence and recommendations. BMJ Open 2016;6:e008859 [Crossref] [PubMed]
- Mate SE, Kugelman JR, Nyenswah TG, et al. Molecular Evidence of Sexual Transmission of Ebola Virus. N Engl J Med 2015;373:2448-54. [Crossref] [PubMed]
- Deen GF, Knust B, Broutet N, et al. Ebola RNA Persistence in Semen of Ebola Virus Disease Survivors - Preliminary Report. N Engl J Med 2015; [Epub ahead of print]. [Crossref] [PubMed]
- Interim advice on the sexual transmission of the Ebola virus disease, 21 January 2016, World Health Organization, Geneva, Switzerland. Available online: http://www.who.int/reproductivehealth/topics/rtis/ebola-virus-semen/en/
Cite this article as: Formenty P, Kieny MP, Broutet N. Perspectives on sexual transmission of Ebola virus: complementarity between research and public health intervention. J Public Health Emerg 2017;1:21.


